

Ildo Nicoletti, Roberta Mannucci, Graziella Migliorati1, Carlo Riccardi1 and Fausto Grignani. Istituti di Medicina Interna e Scienze Oncologiche e di 1Farmacologia, Università di Perugia, 06122 Perugia
Apoptosis is a finely tuned mechanism for the control of cell number in eukariotes. The process is operative during embryogenesis, in tumor regression and in the control of immune response. In most cases, it consists in an ordered sequence of cellular events that start with the transcription of specific genomic sequences, synthesis of specific proteins, and activation of classes of nucleases that cut DNA in the oligonucleosomal subunits. These events are paralleled by specific morphological changes in both the cell nucleus and cytoplasm (Figure 1). Extensive literature exists regarding molecular, biochemical and morphological changes that occurr during apoptosis (for review ref.)
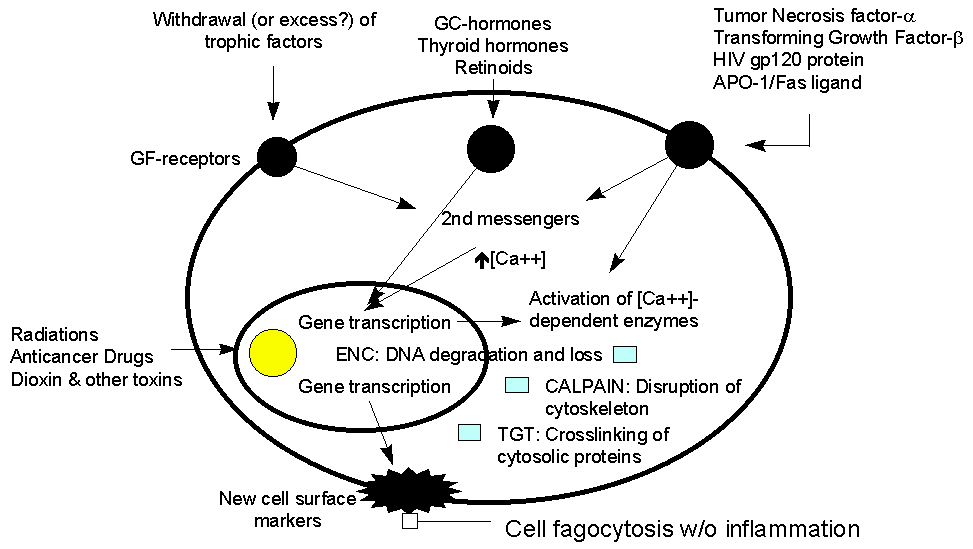
As the majority of the events that characterize apoptotic death can easily be revealed by multiparameter flow cytometry, a lot of methods have been proposed to analyze and quantify the apoptotic process by this type of analysis. In this article we shortly review these techniques and discuss the main advantages and pitfalls of the more common methods.
a. Physical parameters of apoptotic cells
Cells undergoing apoptosis display typical changes in their morphological and physical properties (cell shrinkage, condensation of chromatin and cytoplasm) which are well measurable by flow cytometry. Thus, a series of methods have been proposed for measuring apoptotic cell death through evaluation of light scattering parameters of cells3.
Interaction of a particle with the laser beam produces a light scatter
in a forward direction (FSC, that correlates with cell size) and a lateral
direction (SSC, that correlates with granularity and/or cell density).
While necrotic death is characterized by a reduction in both FSC and SSC
(probably due to a rupture of plasma membrane and leakage of the cells
content), during apoptosis there is an initial increase in SSC (probably
due to the chromatin condensation) with a reduction in FSC (due to the
cell shrinkage). This pattern can be easily observed in some models of
apoptosis, such as the apoptotic death induced by glucocorticoids in murine
thymocytes (Figure 2) but it is much less clear in other cell models (i.e.
the majority of tumor cell lines).
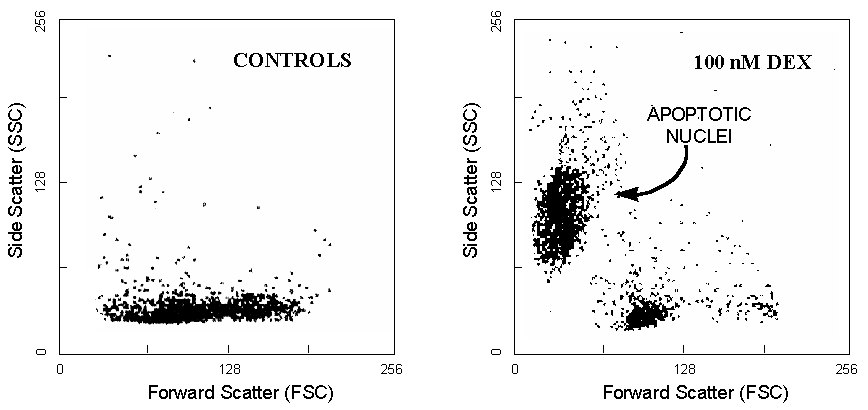
In later stages of apoptosis, furthermore, the intensity of both FSC and SSC decreases due to the emerging secondary necrosis. Therefore, the light-scanner changes are not very specific to apoptosis or necrosis, and light-scanner based methods require accurate controls for eliminating mechanically broken cells, and nuclear debris. This is not a main problem when thymocyte nuclei or T-cells cultured in suspension are analyzed, but is a critical point with cells of myeloid origin or adherent cultured cells.
The main advantages of this analysis are the simplicity, the low-cost and the possibility of its combination with other analysis (i.e. surface immunofluorescence) to identify the phenotype of apoptotic cells in a heterogeneous cell population.
b. DNA content analysis
In 1991 our and other laboratories independently reported that apoptotic cells, but not necrotic ones, have reduced DNA stainability following staining with a variety of fluorochromes. Therefore, the presence of cells with DNA stainability lower than that of G1-cells (hypodiploid or sub-G1 peaks, A0 cells) has been considered a marker of cell death by apoptosis3,.
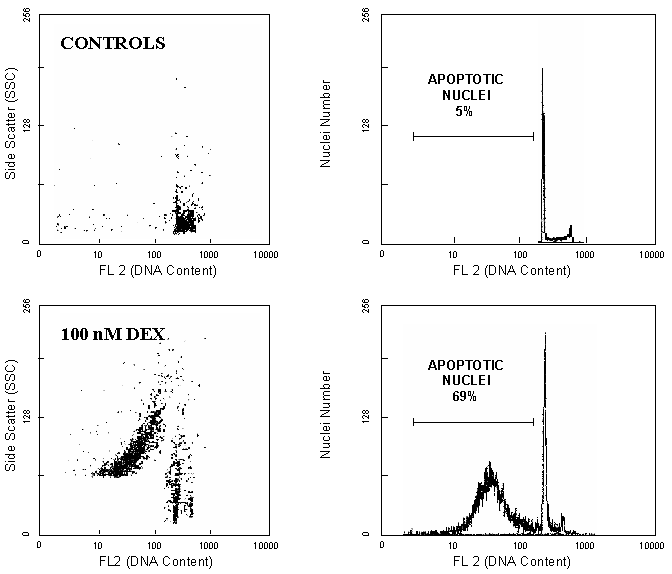
Cellular DNA may be stained with a lot of fluorochromes and methods utilizing propidium iodide, DAPI, Hoechst 33342 and other fluorochromes have been proposed. Since the majority of flow-cytometry are built with a single argon-ion laser (excitation at 488 nm) which does not allow the use of UV-excitable molecules (DAPI and Hoechst 33342) this review will be focused on PI-staining. The reduced stainability of apoptotic cells is the direct consequence of partial loss of DNA due to the activation of endogenous nucleases and diffusion of low-molecular weight DNA outside the cells. This DNA loss is observed when cells are permeabilized by detergents or fixed in ethanol before staining. Whereas the DNA content of unfixed apoptotic cells is unchanged compared to normal cells, detergent treatment releases oligonucleosomes and mononucleosomes. Over 30% of total DNA can be lost as low-molecular weight DNA in apoptotic nuclei. In some situations, when cells are fixed in cold ethanol, immediately stained with PI or DAPI and analyzed with a flow cytometer shortly after staining, the difference in the DNA fluorescence of normal and apoptotic cells is minimal or undetectable. In this cases, a suspension of cells in PBS or hypotonic buffer at the room temperature facilitates the loss of fragmented DNA and produces a clear shift in the sub-diploid peak of apoptotic nuclei.
The appearance of sub-diploid DNA peak is a specific marker of apoptosis;
necrosis induced by metabolic poisons or lysis produced by complement did
not induce any sub-G1 peak in the DNA fluorescence histogram6.
However, in addition to apoptotic cells, the sub-G1 peak can
also represent mechanically damaged cells, cells with reduced chromosome
number (aneuploid cells in a heterogeneous tumor population) and isolated
apoptotic bodies. It is therefore critical that apoptotic cells and debris
are appropriately discriminated. Furthermore, since apoptosis can result
in a complete fragmentation of nucleus and cytoplasm, in some situations
staining of cells in hypotonic buffers can produce several individual nuclear
fragments from a single apoptotic cell. This may result either in a strong
over-extimation of apoptotic cell percentage if isolated apoptotic bodies
are counted as apoptotic cells or in an under-extimation of the phenomenon
if apoptotic cells are gated out as debris. Whilst this is not a significant
problem in thymocytes, since we have demonstrated that both hypotonic staining
solution and alchool fixation before staining produce the same quantitative
results (Figure 4)6, care should always be taken to avoid cell
hyperfragmentation before the flow cytometry analysis.
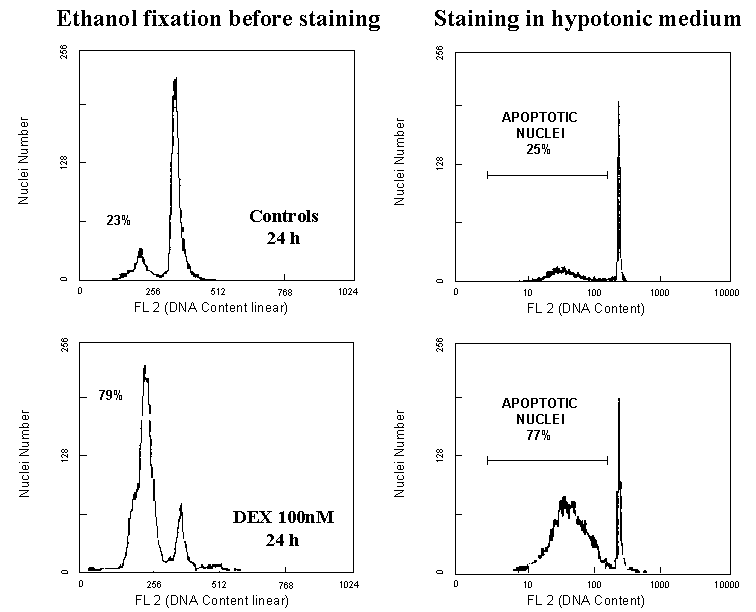
Figure 4: DNA fluorescence flow cytometric profiles of PI-stained thymocytes after 24h incubation in medium alone or medium plus 100 nM DEX. The evaluation of apotosis after either ethanol fixation, RNA-se treatment and PI-staining (DNA fluorescence in linear scale) or after direct PI-staining in hypotonic solution (DNA fluorescence in logarithmic scale) provides very similar results.
In summary, this is a rapid, simple and reproducible method for assessing
apoptosis of specific cell populations, such as thymocytes and tumor cells
of lymphoid lineage. In these models the flow cytometric data show an excellent
correlation with the results obtained with both electrophoretic, microscopic
and colorimetric methods. In other cell models, such as adherent tumor
cell lines and cells of myeloid origin nuclear fragments may be mistakenly
identified as apoptotic cells. In these cases a careful multiparametric
analysis (DNA vs. cell volume and DNA vs. cell density) and a direct microscopic
evaluation of cell suspensions before the flow cytometry are essential
steps for obtaining reproducible results.

Figure 5: Upper panels: TdT assay in HL-60 myeloid cells after 6h treatment with tyrphostin-25. Observation at the fluorescence microscopy reveals incorporation of FITC-dUTP in apoptotic cells (B) but not in normal controls (A). Lower panels: Flow cytometric contour plots of the same experiment. Nuclei are counterstained with PI. This allows a clear extimate of the relationships between cell-cycle position and apoptosis.
Fluorochrome-conjugated avidin or digoxygenin antibodies are used as second step to label the DNA breaks and more recently, fluorochrome-conjugated deoxy-nucleotides have been made available for a single-step labelling procedure. Detection of breaks requires a pre-fixation with a cross-linking agent, such as paraformaldheyde that unlike ethanol prevents the extraction of fragmented DNA. Cells are then post-fixed in cold ethanol, labelled with nucleotides in a TdT catalysed reaction, resuspended in PBS and counterstained with PI for a simultaneous analysis of cell-cycle and apoptosis.
These methods are more expensive and complex when compared to the above reported procedures, but they offer some important advantages in specific situations. A first advantage of the nick translation assay appears to be its ability to reveal early DNA breaks during apoptosis, prior to the loss of any DNA content or morphological variations. A second important advantage is the unique possibility to analyse the cell-cycle position of apoptotic cells, since the DNA content of both normal and apoptotic nuclei can be simultaneously measured. Furthermore, since only apoptosis produces a significant number of DNA breaks (necrotic cells, irradiated cells or cells treated with DNA-damaging drugs have only few breaks) this method appears to be the most specific in terms of early positive identification of apoptotic cells.
e. Expression of antigens and/or specific molecules in apoptotic cells.
Apoptotic cells seem to express specific genes and synthesize a set of specific proteins. Tissue transglutaminase (TTG) is one of these specific markers of apoptosis and it has been clearly demonstrated that immunoistochemical staining for TTG is a good marker of apoptotic death. However TTG staining is of little utility in flow cytometry, since the cytosolic localization of the antigen leads to a lot of specificity problems.
Another important characteristic of apoptotic cells is their early recognition by macrophages and phagocytes before they have lost membrane integrity. Wyllie et al. demonstrated that this is due to a change in the membrane structure of apoptotic cell, which a macrophage-associated lectin could recognize. More recent studies provided convincing evidence to suggest that phosphatidylserine (PS) which is normally confined to the inner leaflet of the plasma membrane is externalized in the early phases of apoptosis. This has ben confirmed in a number of apoptosis models and it has been shown that inhibition of apoptosis, by overexpression of repressor proteins bcl-2 or abl, also inhibits PS exposure on cell surface. In apoptotic cells the externalized PS can be readily detected not only by macrophage lectins but also by FITC-coupled annexin-V. Annexin-V, a Ca++-binding protein is able to interact with negatively charged phospholipids, such as PS, in a Ca++-dependent manner. Thus, apoptotic cells exposing PS can be identified and measured at the both fluorence microscope and flow cytometer after FITC-Annexin-V staining.
An important advantage of this method is that no fixation or permeabilization step is required. Cells are simply incubated in a buffer containing annexin-V conjugated to FITC and Ca++ions, allowed to bind for 5-10 minutes, and analysed immediately. An other important advantage is that PS externalization seems to occur very early in apoptosis, before any nuclear changes have occurred. Therefore, as reported for TdT assay, this technique should be preferred when initial phases of the apoptotic process are investigated.
Conclusions:
Flow cytometry can be applied in basic research and in the clinic to identify and measure apoptotic cells. The choice of a particular flow cytometry method depends on several variables (cell system, type of flow cytometer, type of apoptosis, type of apoptosis inducer, type of information required). Regardless of the flow cytometry technique used to measure apoptosis, in most situations the type of cell death should be confirmed by direct microscope inspection. Apoptotic cells display a very specific pattern of morphological changes at the light, electron and fluorescence microscope and this should be the deciding factor when ambiguity arises regarding the mechanism of cell death. It should also be stressed that apoptosis is a sequential process of variable (1-8 h) duration, depending on inducer and cell type. Any method has an optimal time window for analysing the apoptotic process and it is possible that the percentage of apoptotic cells in the same population may not be identical when two methods are utilized (i.e. Annexin-V and TdT-assay are more sensitive than DNA-content analysis in the early phases of apoptosis). The type of information that is being sought is also critical. For example, if specificity of apoptosis with respect to cell-cycle phase is investigated, TdT-assay with PI counterstaining is the method of choice.
In conclusion, despite no single method can provide unequivocal quantitative
measure of apoptosis in all situations, flow cytometry techniques represent
very useful tools for a variety of biological and clinical studies requiring
quantitative information about cell death.
APPENDIX
1. Analysis of scatters parameters of apoptotic cells by flow cytometry
a. Quick method (direct DNA staining in fluorochrome solution)
1. Wyllie AH, Kerr JFR, Currie AR. Cell death: the significance of apoptosis. Int Rev Cytol 68:251-307, 1980
2. Hale AJ, Smith CA, Sutherland LC, Stoneman VEA, Longthorne VL, Culhane AC, Williams GT. Apoptosis: molecular regulation of cell death. Eur J Biochem 236:1-26, 1996
3. Darzynkiewicz Z, Bruno S, Del Bino G, Gorczyca W, Hotz MA, Lassota P, Traganos F. Features of apoptotic cells measured by flow cytometry. Cytometry 13:795-808, 1992
4. Migliorati G, Nicoletti I, Pagliacci MC, DAdamio L, Riccardi C. Interleukin-4 protects double-negative and CD4 single-positive thymocytes from dexamethasone-induced apoptosis. Blood 81:1352-1358, 1993
5. Swat W, Ignatowicz L, Kisielow P. Detection of apoptosis of immature CD4+8+ thymocytes by flow cytometry. J Immunol Meth 137:79-87, 1991
6. Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C. A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Meth 139:271-279, 1991
7. June CH. Analysis of lymphocyte activation and metabolism by flow cytometry. Curr Opinion Immunol 4:200-204, 1992
8. Dive C, Gregory CD, Phipps DJ, Evans DL, Milner AE, Wyllie AW. Analysis and discrimination of necrosis and apoptosis (programmed cell death) by multiparameter flow cytometry. Bioch Biophys Acta 1133:275-285, 1992
9. Zamai l, Falcieri E, Zauli G, Cataldi A, Vitale M. Optimal detection of apoptosis by flow cytometry depends on cell morphology. Cytometry 14:891-897, 1993
10. Gavrieli Y, Sherman Y, Ben-Sasson SA. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119:493-501, 1992
11. Gorczyca W, Gong J, Darzynkiewicz Z. Detection of DNA strand breaks in individual apoptotic cells by the in situ terminal deoxynucleotidyl transferase and nick translation assays. Cancer Res 53:1945-1951, 1993
12. Rogaia D, Grignani F, Grignani F, Nicoletti I, Pelicci PG. The acute promyelocytic leukemia-specific PML/RARa fusion protein reduces the frequency of committment to apoptosis upon growth-factor deprivation of GM-CSF dependent myeloid cells. Leukemia, 9:1467-1472, 1995
13. Piacentini M, Fesus L, Farrace MG, Ghibelli L, Piredda L, Melino G. The expression of tissue transglutaminase in two human cancer cell lines is related with the programmed cell death (apoptosis). Eur J Cell Biol 54:246-254, 1991
14. Duvall E, Wyllie AH, Morris RG. Macrophage recognition of cells undergoing programmed cell death (apoptosis). Immunology 56:351-358, 1985
15. Fadok VA, Voelker DR, Campbell PA, Cohen JJ, Bratton DL, Henson PM. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol 148:2207-2213, 1992
16. Koopman G, Reutelingsperger CPM, Kuijten GAM, Keehnen RMJ, Pals ST, van Oers MHJ. AnnexinV for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis. Blood 84:1415-1420, 1994
